More than 3,000 years ago, ancient Egyptian and Greek texts already referenced the healing properties of sunlight. Herodotus, the founder of heliotherapy, described the benefits of sunlight in treating vitiligo and psoriasis, and its preventative effect on rickets and depressive psychoses.
Dr. Niels Ryberg Finsen's studies on the effects of sunlight on cutaneous tuberculosis, which led to his being awarded the Nobel Prize in Medicine in 1903, culminated in his receiving this award. He demonstrated a bactericidal effect of solar radiation along with a regenerative effect on tissues. He also warned of the harmful effects of excessively high doses of radiation on scars, inducing hyperpigmentation secondary to cellular damage in smallpox.
In 1967, Hungarian physician Dr. Endre Mester, a few years after the first laser devices were invented, wanted to study whether laser radiation could cause skin cancer in mice. He shaved the backs of mice and then applied laser treatment with a low-power 694nm ruby laser. There were no cases of cancer, but he observed that hair grew back more quickly in the mice treated with the low-power laser than in the untreated control group. This was the first practical demonstration of laser biostimulation.
Subsequently, multiple studies in cell cultures, animal models, and humans have demonstrated that the response to light therapy is inversely dose-dependent: that is, low doses of light energy have a regenerative effect on cells, while high doses can have an inhibitory effect on the cellular response. For this reason, the English term LLLT (low-level light therapy) was coined to describe these therapies in which low doses of energy are converted into metabolic energy, subsequently modulating biological functions, as we will see in the next section. Therapy light of low intensity acts by a mechanism of photobiomodulation.
Starting in 1960, NASA researchers began studying the effects of low-intensity light therapy on astronauts subjected to the effects of weightlessness in space. Weightlessness causes slowed wound healing and muscle atrophy in astronauts, effects that could be partially mitigated with light therapy.
Low-intensity light therapy is currently used in a wide range of clinical situations. In neonatal jaundice, 460nm blue light is used to accelerate bilirubin breakdown. Light therapy is also currently used in sports medicine, inflammatory joint diseases, neurological disorders, scar healing and skin regeneration, and inflammatory skin diseases.
WHAT IS LOW INTENSITY LIGHT THERAPY?
Light is a type of electromagnetic radiation that, according to modern quantum physics, consists of both particles (photons, which are particles or quanta of energy) and waves. The energy of photons depends on the wavelength of the electromagnetic radiation. When photons interact with living tissue, they can be absorbed or reflected. Absorbed photons interact with organic molecules or chromophores, exciting them and causing an electron jump in the peripheral electrons of these chromophores. This energy change in cells is used to alter cellular functions. Low-intensity light therapy works by converting light energy into metabolic energy, with the subsequent modulation of cellular biological function. For this reason, low-intensity light therapy is called photobiomodulation. In this respect, it is important to distinguish this therapy from the effects of high-power lasers, in which the tissue absorbs high energies resulting in heating and cell destruction. The energy doses are not high enough to cause heating and cell destruction, but they are sufficient to modulate cellular functions.
Sunlight is polychromatic, meaning it's a mixture of different wavelengths, ranging from ultraviolet waves, which have shorter wavelengths and penetrate tissue less deeply, to infrared waves, which have longer wavelengths and greater penetrating power. Low-intensity light therapy uses lasers or LEDs (light-emitting diodes) of a specific wavelength that is absorbed by a chromophore, or specific cellular structure, triggering the modulation of biological functions. Lasers are monochromatic, meaning they emit light of a very specific wavelength in a coherent manner (the waves are emitted simultaneously, exhibiting spatial and temporal synchronization). This has the advantage of increasing tissue penetration. LEDs, on the other hand, emit nearly monochromatic light within a narrow wavelength range, typically between 4 and 10 nm. This light is incoherent, meaning waves of the same wavelength are emitted at different times. The incoherence of LED light can be advantageous in low-intensity light therapy because it allows tissue exposure to therapeutic wavelengths at low energy densities for a sufficient duration to modulate cellular metabolism. Another significant advantage of LEDs is their ability to treat much larger areas than lasers. Low-intensity light therapy utilizes wavelengths from the visible spectrum without employing the ultraviolet radiation present in sunlight, which is responsible for skin cancer.
In the past, there had been conflicting results regarding the effectiveness of low-intensity light therapy because either energy densities that were too low or too high, irradiances measured in Watts per cm² that were too high, exposure times, or wavelengths that were unsuitable. Currently, there is sufficient evidence that if the appropriate light parameters are used, based on the physicochemical properties of the tissue in question, the therapy is effective.
It is very important to keep in mind that the effectiveness of the therapy in relation to the energy doses used follows an inverted U-shape. That is, minimum doses are needed to stimulate the cell, but as the dose increases, there comes a point where the effect reverses and an inhibitory effect occurs, as can be seen in the graph.
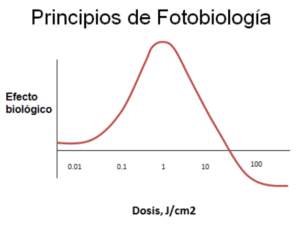
The most effective energy doses range between 1 and 7 J/cm2 with irradiances of 5-100 mW/cm2.
MECHANISM OF ACTION
Nature has designed systems to harness electromagnetic radiation for the maintenance of biological functions. The use of electromagnetic radiation is fundamental for the maintenance of life on Earth. Light energy emits photons that, when interacting with certain cellular biomolecules, excite them and thus trigger metabolic processes. Within cells there are chromophores that will absorb photonic energy. A chromophore is a molecule or part of a molecule that gives a specific color to the compound it is part of and that will interact with light energy. Examples of chromophores are chlorophyll (used by plants for photosynthesis), hemoglobin in red blood cells, cytochrome c oxidase (the mitochondrial enzyme responsible for converting ADP into ATP used in the Krebs cycle to generate energy), myoglobin in muscles, flavoproteins, and porphyrins. It is interesting to note that all these chromophores share a basic chemical structure similar to that of protoporphyrin. The diagram shows how electromagnetic radiation interacts with chlorophyll in the case of plants and with the mitochondrial enzyme cytochrome c oxidase in the case of animals to stimulate a photochemical reaction that will culminate in photosynthesis in the case of plants and in the mitochondria of animal cells in the generation of energy in the form of ATP.
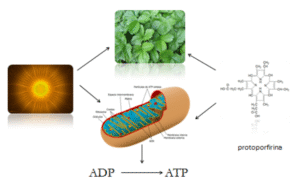
Red or infrared wavelengths are those that produce the greatest biological effect due to their greater absorption by mitochondrial cytochrome c oxidase.
Studies by Tina Karu and subsequent work have demonstrated that low-intensity light therapy, in addition to increasing cellular ATP, alters the cell's redox potential. This change, in turn, signals the activation of nuclear gene expression, leading to the production of growth factors, cytokines, and enzymes. Light has been found to activate 111 genes. In simplified terms, light activates the mitochondrial enzyme cytochrome oxidase, causing a change in redox potential that stimulates both ATP production and, at low doses, the generation of free radicals. The increase in ATP results in an increase in cAMP and calcium, which, along with free radicals, act as second messengers, stimulating gene expression at the nuclear level. These genes are responsible for the formation of growth factors, anti-inflammatory cytokines, decreased apoptosis (cell death), and increased or decreased activity of various enzymes. So, will light stimulate cellular functions in all cells? No, only cells with a reduced redox state (low or acidic intracellular pH) will respond markedly, while cells in an optimal redox state will not. Therefore, the mechanism of action of this therapy is called photomodulation because it acts by regulating altered cellular functions.
CLINICAL APPLICATION OF LOW INTENSITY LIGHT THERAPY.
We have observed that light therapy through photobiomodulation regulates metabolism, cell migration, cell proliferation, cellular inflammation, and the synthesis and secretion of various proteins. These properties have been used clinically to stimulate tissue regeneration, treat skin aging, and reduce inflammation and pain.
- WOUND HEALING AND TISSUE REGENERATION
Low-intensity light therapy promotes wound healing by first stimulating the production of pro-inflammatory cytokines present in the initial phase of healing. Secondly, it stimulates the proliferation and migration of fibroblasts. Thirdly, it stimulates the secretion of vascular growth factors responsible for neovascularization and growth factors that induce collagen formation by fibroblasts. Finally, it induces the transformation of fibroblasts into myofibroblasts, which are the cells responsible for wound contraction.
- SKIN REJUVENATION
Barolet's studies and numerous subsequent studies support the usefulness of low-power LEDs in the treatment of skin aging. They work by promoting collagen synthesis by dermal fibroblasts on the one hand, and by inhibiting the breakdown of collagen on the other, because the LEDs inhibit the production of metalloproteinases, enzymes responsible for breaking down collagen. Furthermore, the deposition of new collagen is encouraged, forming an organized network beneath the epidermis that is clearly different from the small, thin, disorganized collagen that characterizes aged and sagging skin. Low-intensity light therapy is beneficial when used after more aggressive treatments such as chemical peels, laser treatments, injections, and surgeries because, thanks to its anti-inflammatory effect, it can help avoid side effects and promote skin regeneration, preventing complications. One of the immediate effects that appears after a photobiomodulation session is an increase in skin luminosity, an effect highly appreciated by patients.
- INFLAMMATORY SKIN DISEASES.
Along with appropriate medical treatment, light therapy can be helpful in various inflammatory conditions due to its anti-inflammatory effect. In rosacea, a chronic inflammatory disease characterized by redness and pustular lesions, light therapy, in conjunction with appropriate medical treatment, has an anti-inflammatory effect and regulates blood and lymphatic circulation.
- HYPERPIGMENTATION AND MELASMA
In cases of post-inflammatory hyperpigmentation and melasma (facial pigmentation of probable hormonal origin), low-intensity light therapy is useful in regulating melanocytic function along with the appropriate dermatological treatment in each case.
- PHOTOPROPHYLAXIS
Barolet's studies demonstrated that in normal patients and in patients with polymorphic light eruption (solar hypersensitivity), 10 treatments with low-intensity red LEDs (660nm) provided sun protection equivalent to a sun protection factor (SPF) of 15 and a decrease in post-inflammatory hyperpigmentation after increasing doses of ultraviolet radiation exposure. Previously, Menzes' studies had demonstrated in vitro that infrared radiation protects dermal fibroblasts from the toxicity of solar ultraviolet rays. The mechanisms involved include effects at the level of nuclear DNA, blocking the effects of ultraviolet radiation on genes such as p53 (which induces apoptosis or cell death) and on genes that regulate the secretion of proopiomelanocortin, from which melanin-stimulating hormone (MSH) is derived, in addition to a direct effect on melanocytes, regulating the melanin formation.
- ACNE
LED therapy has been used in acne treatment due to the effects of blue light, which is absorbed by Propionibacterium acnes, the bacteria involved in acne, causing its destruction. This effect is photodynamic and requires higher energy doses. Red light is added to the blue light to treat inflammation. Acne treatment should always be accompanied by dermatological treatment and monitoring.
- ANDROGENETIC ALOPECIA AND HAIR LOSS
Recent studies support the usefulness of low-intensity light therapy in various types of alopecia. Studies in mice and humans demonstrate that the therapy is able to stimulate hair bulb stem cells and regulate the hair cell cycle, inducing the transition of hairs from a resting state (catagen and telogen) to an active growth state (anagen).
- PHOTODYNAMIC THERAPY. TREATMENT OF SKIN PRECANCER.
I would like to briefly mention photodynamic therapy, which uses the same LED devices as low-intensity photomodulation therapy but at higher doses, resulting in the destruction of atypical cells. In this latter case, a photosensitizing cream is applied before irradiation, which is preferentially absorbed by the atypical cells. After an incubation period, the affected area is irradiated with red light at 37 J/cm², inducing a photochemical reaction that selectively destroys the atypical cells. This treatment is approved for actinic keratoses (erythematous-desquamative lesions that appear on photoaged skin and are atypical, presenting a risk of developing into skin cancer) and for certain types of superficial skin cancer.
- OTHER NON-DERMATOLOGICAL INDICATIONS FOR LOW-INTENSITY LIGHT THERAPY
Recent research has demonstrated the beneficial effects of low-intensity light therapy in optic nerve trauma, methanol poisoning, optic neuropathy, retinal injury, retinitis pigmentosa, phototoxicity, macular degeneration, anoxic brain injury, stroke, neurodegenerative diseases, cognitive impairment, depression, pain management, sports medicine, and rehabilitation. In all these cases, the same concept of photomodulation is used to provide more energy to cells and regulate cellular regenerative functions.
KEY WORDS:
Low-intensity light therapy, photobiomodulation, regulation of cellular functions, anti-inflammatory, luminosity, skin rejuvenation, inflammatory skin diseases, post-inflammatory hyperpigmentation, melasma, sun protection, acne, hair loss, androgenic alopecia
CONCLUSION
Low-intensity light therapy, also called photobiomodulation, is a safe and effective tool that works by increasing cellular energy. It acts at different levels: mitochondrial activation, regulation of nuclear gene expression, on the redox potential of cell membranes, and as an anti-inflammatory. It has an immediate effect of increasing skin radiance and is combined with other treatments (in skin rejuvenation, inflammatory skin diseases, post-inflammatory hyperpigmentation, melasma, scarring, burns, acne, sun protection, androgenetic alopecia, and hair loss), constituting a cornerstone of modern integrative dermatology.
BIBLIOGRAPHY
- Mester E, Szende B, Gärtner P. The effect of laser beams on the growth of hair in mice. Radiobiol Radiother (Berl). 1968;9(5):621-6. PMID: 5732466.
- Finsen NR. Om lysets Indvirkninger paa Huden [On the effects of light on the skin]. København: Finsen; 1893. (Historical work; Finsen won the Nobel prize in 1903 for treatment of the light in lupus vulgaris). Historical source: Nobel Prize / historical revisions.
- Maghfour J, Ozog DM, Lim HW, Kohli R, Jagdeo J. Photobiomodulation CME Part I: Overview and mechanism of action. J Am Acad Dermatol. 2024. PMID: 38309304.
- Mineroff J, Maghfour J, Ozog DM, Lim HW, Kohli R, Jagdeo J. Photobiomodulation CME Part II: Clinical applications in dermatology. J Am Acad Dermatol. 2024. PMID: 38307144.
- Hernández-Bule ML, Naharro-Rodríguez J, Bacci S, Fernández-Guarino M. Unlocking the Power of Light on the Skin: A Comprehensive Review on Photobiomodulation. Int J Mol Sci. 2024;25(8):4483. doi:10.3390/ijms25084483. PMID: 38674067; PMCID: PMC11049838.
- Glass GE, Haxton $ C, et al. Photobiomodulation: A systematic review of the oncologic safety of low-level light therapy for aesthetic skin rejuvenation. Aesthet Surg J. 2023;43(5):NP357–NP371. doi:10.1093/asj/sjad018. PMID: 36722207; PMCID: PMC10309024.
- Oliveira S, Andrade R, Valente C, Espregueira-Mendes J, Silva FS, Hinckel BB, et al. Effectiveness of photobiomodulation in reducing pain and disability in patients with knee osteoarthritis: a systematic review with meta-analysis. Phys Ther. 2024;104(8): (article). doi:10.1093/ptj/pzae073. PMID: 38775202.
- Ji Q, Yan S, Ding J, Zeng X, Liu Z, Zhou T, et al. Photobiomodulation improves depression symptoms: a systematic review and meta-analysis of randomized controlled trials. Front Psychiatry. 2024;14:1267415. doi:10.3389/fpsyt.2023.1267415. PMID: 38356614; PMCID: PMC10866010.
- Cheng YP (Fan SMY?), et al. A 24-Week, Randomized, Double-Blind, Self-Comparison, Sham-Controlled Trial of Low-Level Light Therapy for Androgenetic Alopecia. [Journals—publication record] 2018; (24-week RCT). PMID: 29957664. (Clinical trial pivotal on LLLT and AGA).
- Kim H-S, Lim H-W, et al. Low-level laser therapy for androgenetic alopecia: randomized trial (24-week). Dermatol Surg. 2013;39(4): (detail in PubMed). PMID: 23551662.
- Jorge AES, Dantas WHAT, Aburquerque-Sendín F, et al. Photobiomodulation does not provide incremental benefits to patients with knee osteoarthritis who receive a strengthening exercises program: a randomized controlled trial. Braz J Phys Ther. 2023 Jul-Aug;27(4):100519. PMID: 37572382; PMCID: PMC10440468.
- Zhou Y, et al. Efficacy of low-level light therapy for improving healing of diabetic foot ulcers: a systematic review and meta-analysis of randomized controlled trials. Wound Repair Regen. 2021 Jan;29(1):34-44. doi:10.1111/wrr.12871. PMID: 33078478.
- Dompe C, Moncrieff L, Matys J, Grzech-Lewandowska M, et al. Photobiomodulation—underlying mechanism and clinical applications. J Clin Med. 2020;9(6): (review). PMID/PMCID: PMC7356229 (full review on PMC).
- Vassão PG, Ribeiro E, et al. Effects of photobiomodulation and a physical exercise program on biomarkers and functional capacity in women with knee osteoarthritis: randomized clinical trial. Adv Rheumatol (São Paulo). 2021; study (RCT). PMID: 34656170.
- Pauli Paglioni M, Araújo ALD, Grove LPA, et al. Tumor safety and side effects of photobiomodulation therapy used for prevention and management of cancer treatment toxicities: systematic review. Oral Oncol. 2019;93:21-28. doi:10.1016/j.oraloncology.2019.04.004. PMID: 31109692.
- Mineroff J, et al. Cutaneous effects of photobiomodulation with 1072-nm light: clinical observations and safety. J Invest Dermatol. 2023. PMID: 36495337.
- Galache TR, Seine MM, Pavani C, et al. Amber photobiomodulation versus tranexamic acid for the treatment of melasma: protocol for a double-blind, randomised controlled trial. BMJ Open. 2023;13(7):e073568. doi:10.1136/bmjopen-2023-073568. PMID: 37479524; PMCID: PMC10364183.
- Glass GE; World Association for Laser Therapy (discussion). Evidence-based consensus on the clinical application of PBM: expert panel conclusions (dermatology-focused consensus). Consensus/Review. 2024-2025 (panel report / CME summary). PMID/record: 40253006 (consensus summary).
- Mendes-Costa, LDS, et al. Photobiomodulation: systematic review and meta-analysis of parameters used in the resolution of diabetic foot ulcers / wound healing — parameter guidance. J Clin Transl Res / Wound Repair journals. 2020-2021. (summary and meta-analyses). PMID record series (see PubMed 33190161 / 33078478).
- Jean-Pierre P, et al. Lasers in the management of alopecia: review (includes translation and citation of Mester 1968 and modern RCTs). J Cosmet Dermatol / Photomed Laser Surg. 2024; (review; PMC open access). PMID/PMCID listed in PubMed Central.

